PHOTOBIOMODULATION THERAPY
FOR DRY AMD
The First and Only FDA-Authorized Treatment for Early to Intermediate Dry Age-Related Macular Degeneration
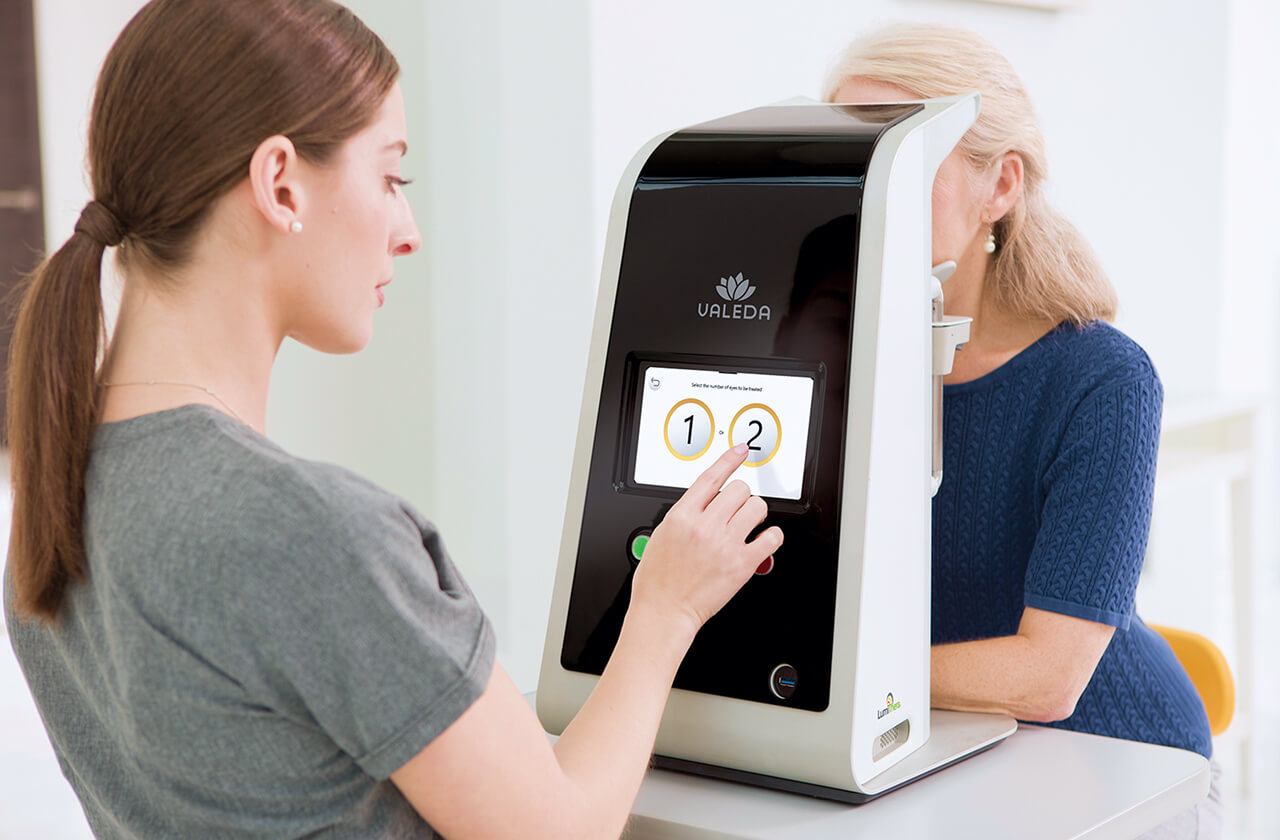
Valeda Light Therapy

To qualify for the Valeda treatment, patients must undergo an evaluation which includes dilation and retinal imaging by a retina specialist at California Retina Consultants to confirm eligibility.
If eligible, a dedicated patient consultant will work with you closely from evaluation to treatment!
About Age-Related Macular Degeneration
Age-related macular degeneration (AMD) is a leading cause of vision impairment in older adults. This condition occurs when the cells in the macula, the central part of the retina, begin to deteriorate and lose their effectiveness. As AMD progresses, symptoms may include visual distortion, blurred central vision, difficulty with reading or tasks that require close-up work, and challenges adjusting to changes in light.
AMD is classified into two types: dry and wet. Dry AMD is the more common form. The severity of dry AMD can range from mild to severe, but vision decline typically occurs more slowly compared to wet AMD.
To learn more about age-related macular degeneration (AMD), click here.
How the Treatment Works
- Enhancing blood circulation to the retina and its supporting structures
- Promoting the production of nitric oxide, a vital compound for the health of blood vessels
- Activating mitochondria, the tiny particles within cells responsible for energy generation
The Valeda® device is the first specifically created for the treatment of Dry AMD.
What to Expect

- Nine sessions, with two to three treatments per week for three to five weeks.
- Each session lasts just a few minutes and utilizes three different wavelengths of light.
- Subsequent treatment rounds are recommended every four to six months, as advised by your retina specialist.
Patients typically report no discomfort or side effects from Valeda® treatments and can resume normal activities after their sessions. You might experience a brief afterimage, which should fade within a few minutes. Treatment sessions require minimal preparation.
Treatment can be performed in one eye (unilateral) or both eyes (bilateral).
The pricing is as follows:
- $3,000 per series for bilateral treatment (OU) (9 treatments per series)
- $2,400 per series for unilateral treatment (one eye) (9 treatments per series)
Ask us about the savings available with our annual treatment packages. Contact our team today for more details.
Insurance Coverage
Coverage for photobiomodulation therapy varies by carrier. Our team will verify benefits and discuss financial responsibility prior to treatment.
The Clinical Research Behind Light Therapy
Three multicenter clinical studies, known as LIGHTSITE I, II, and III, were conducted at top retinal centers nationwide to evaluate the safety and effectiveness of the now FDA-authorized Valeda® Light Delivery System. Participants in these trials were individuals diagnosed with early to intermediate stages of dry AMD.
The LIGHTSITE Phase III trial lasted 24 months and enrolled 148 eyes from 100 participants who received either Valeda® PBM or a placebo (sham) treatment, delivered three times per week for several weeks at a time. Trial findings revealed that in the PBM group, approximately 55% of patients improved by at least five letters on the eye chart at the 13-month mark. This improvement was sustained at the 24-month mark. In contrast, the placebo group showed minimal improvement.
In addition to improvements in vision, participants in the LIGHTSITE Phase III trial receiving PBM treatment demonstrated a stabilization of drusen, the fatty deposits typically linked to dry AMD, while those given a placebo experienced an increase in these deposits.
Participants in the PBM trial also showed a decreased likelihood of advancing to severe stages of AMD. There were fewer instances of wet AMD and geographic atrophy (GA)—a severe form of dry AMD that results from the degradation of macular cells and can lead to permanent vision loss—in those treated with PBM. By hindering or delaying progression to these more advanced stages, patients may preserve their vision for a longer period.
For a closer look at the science behind Valeda®, visit the LumiThera website.
Indications for Use

Ideal patients are those who exhibit signs of early dry AMD, specifically the grades deemed appropriate for nutritional supplements, and have a visual acuity of 20/70 or better.
- Patients with early to intermediate dry age-related macular degeneration may be candidates. Eligibility is determined by a retina specialist following consultation and imaging. Valeda is not intended for wet AMD and may not be recommended for certain advanced forms of dry AMD.
Safety
When delivered to the retina, the Valeda® Light Delivery System technology is generally safe with minimal side effects. Studies report no damage to the eye or its photoreceptors, but some individuals may experience mild discomfort, eye strain, irritation, or temporary vision changes during or after treatment. However, these symptoms are rare and typically resolve quickly.
Clinical trials are ongoing, but current evidence suggests that Valeda® photobiomodulation is a powerful, low-risk treatment option for patients with AMD, especially in the absence of alternative effective treatments.
Contraindications
As a precaution, patients should not be treated with Valeda® if they have known photosensitivity to yellow or red light, near-infrared radiation, or a history of light-activated central nervous system disorders, such as seizures, epilepsy, or migraines.
Additionally, patients should not receive treatment within 30 days of using photosensitizing agents, such as topicals or injectables that are affected by light at 590, 660, or 850 nm, without first consulting a physician at California Retina Consultants.
Request an Appointment at California Retina Consultants
Valeda light therapy is currently only offered at our Santa Barbara and Valencia offices.
If you have Photobiomodulation, please schedule a consultation with us to learn more about Valeda® and determine whether you qualify for this treatment. If you’re already a patient with us, you can discuss this option with your doctor at your next appointment.
Santa Barbara:
525 East Micheltorena St,
Suite A,
Santa Barbara, CA 93103
Phone: 805.963.1648
Fax: 805.965.5214
Valencia:
23501 Cinema Drive
Suite 109
Valencia, CA 91355
Phone: 661.253.2939
Fax: 661.253.0643
